News
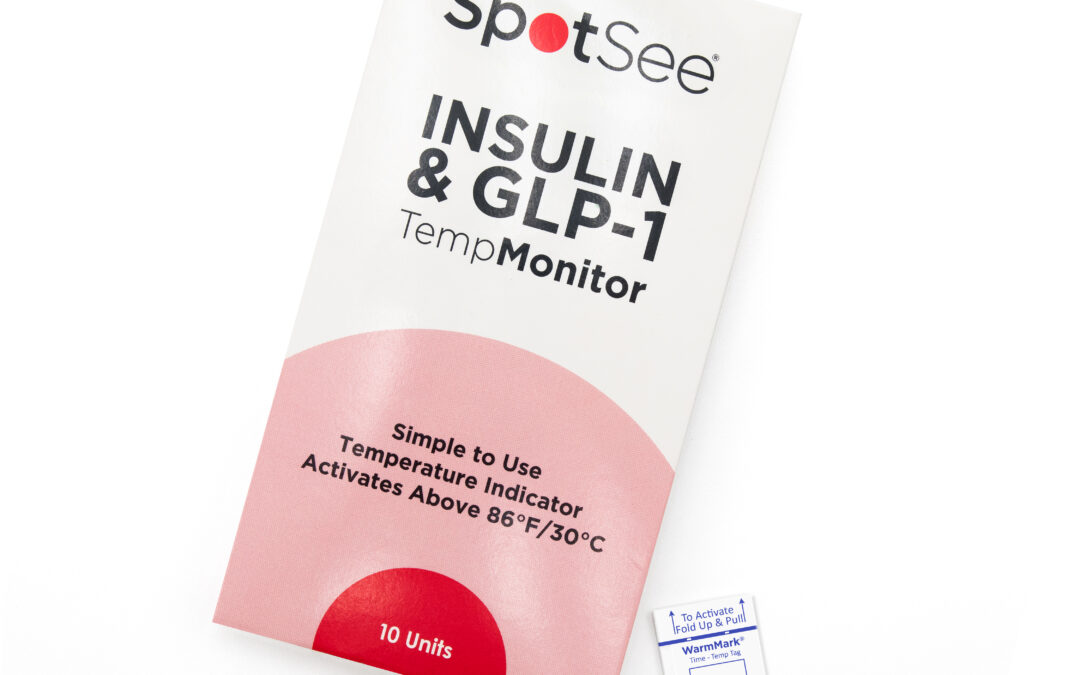
SpotSee Launches TempMonitor to Track Temperature Integrity of Insulin and GLP-1 Medications
SpotSee, a global leader in condition-indicating and monitoring solutions, today announced the launch of TempMonitor™, a simple and affordable single-use temperature indicator designed specifically for patients who rely on insulin and Glucagon-like, peptide-1...

SpotSee ColdChain Complete: The cold chain monitoring solution
What you need to know about cold chain monitoring and how SpotSee ColdChain Complete can help Key Takeaways Understanding why cold chain monitoring is important with international logistics. Knowing the challenges of international cold chain monitoring Seeing how...

Mitigate the Risk – Safeguard Your Energy Infrastructure During Transport
Get the detailed transport insights you need to prevent damage and improve the bottom line Key Takeaways: You take a risk every time you ship power-generation components. Built with narrow tolerances, they require sensitive handling to avoid damage. Preserving product...

SpotSee Branding and Product Updates – March 2024
If you've been keeping an eye on SpotSee lately, you might have noticed a few changes happening. We are excited to share some updates on our branding and product lineup that we think you'll find interesting. A Fresh Look for SpotSee First things first – we've given...

How Safe is Your Blood Bag Supply? How to Monitor Blood Bags in Hospitals
More than safety is at stake. Stay compliant and reduce waste with HemoTemp II. Key Takeaways: U.S. blood supplies are at an all-time low. Improper storage leads to 200,000 to one million wasted units annually, costing up to $230 million. Patient safety is at risk –...

SpotBot® 4G Plus Impact: Analyzing the Ripple Effects of Impact Damage
How to Tackle Impact Damage Key Takeaways: Learn what causes impact damage. How SpotBot 4G Plus technology and functionality help prevent damage. Tips for using monitoring data to analyze the effects of impact. Businesses that ship products need effective...

Unraveling the Roots of Product Damage in the Supply Chain: A Comprehensive Guide
Key Takeaways: What are the common causes of product damage? Best practices for monitoring product journeys to prevent damage. Strategies for supply chain continuous improvement. Emerging technology applications for improving the supply chain. Product Damage Risk...

Delving into Precision: Deep Dive of Temperature Monitoring with Data Loggers
Achieve Accuracy and Compliance with Data Loggers Key Takeaways: How to achieve temperature monitoring accuracy and compliance with data loggers. What are data loggers, and how do they work? Discover the benefits of using data loggers for monitoring shipments. Learn...

Navigating Temperature Sensitivity: Effects of Temperature Change on Biologic Drugs
How Do Temperature Changes Affect the Stability of Biologic Drugs? Let’s Find Out. Key Takeaways: Why are biologic drugs so sensitive to temperature and impact? What is the impact of temperature fluctuations on biologic drugs? How can temperature be controlled and...

Understanding Distinctions in FMD (EU) and DSCSA (US) Regulations
Navigating the Differences and Current State of the FMD and DSCSA Key Takeaways: Comprehensive explanations of FMD and the DSCSA How do FMD and DSCSA compare? Find out what FMD and DSCSA have in common and how they differ. Regulatory compliance laws for the FMD...

Navigating the Future Innovations of Cold Chain Compliance
Cold Chain Technology and Compliance: Insights for the Future. Key Takeaways: What are the recent technological advancements in cold chain monitoring? Cold chain regulatory compliance changes and their impact on operations. Importance of proactive planning for cold...
Protecting Flu Vaccines Using WarmMark® Time-Temperature Indicators
Flu vaccines are temperature sensitive biologics that require precise cold chain management to maintain efficacy. Explore how to enhance flu vaccine monitoring, regulatory compliance, distribution safety, and more. Key takeaways: Precise cold chain management is vital...

Adapting to Changing Demands by Leveraging Rail Transportation Advancements
Explore the challenges and opportunities in rail transportation and how Thermax® Temperature Indicators give you an edge. Key Takeaways: The transportation industry is responsible for the highest proportion of global CO2 emissions. Rail produces fewer emissions than...

Understanding the Drug Supply Chain Security Act (DSCSA) – Updates of 2023
Compliance insights for pharmaceutical, diagnostics, pharmacies, third-party logistics (3PLs), supply chain, transportation, and life science companies. Key Takeaways: A new requirement for tracking at the package levels was due for implementation on November 27,...

Keeping Do-Not-Freeze Injectable Drugs Safe Through the Last Mile of Their Journey
Temperature excursions jeopardize a drug's efficacy and pose serious health risks to patients and financial repercussions for pharmaceutical companies. Discover how to ensure do-not-freeze injectable drug stability during last mile distribution. Key Takeaways:...

WarmMark: Last Mile for Pharmacy Home Delivery
SpotSee’s WarmMark® plays a crucial role in identifying temperature excursions during the delivery of prescription medicine to homes. It is a specialized temperature indicator designed to provide clear and actionable information about whether a medication's...

Shock Monitoring for Aerospace Components
Monitoring shock when transporting a satellite from assembly to the launch pad is crucial for several important reasons: Preservation of Equipment Integrity: Satellites are highly sensitive and complex pieces of equipment with delicate internal components. Even minor...
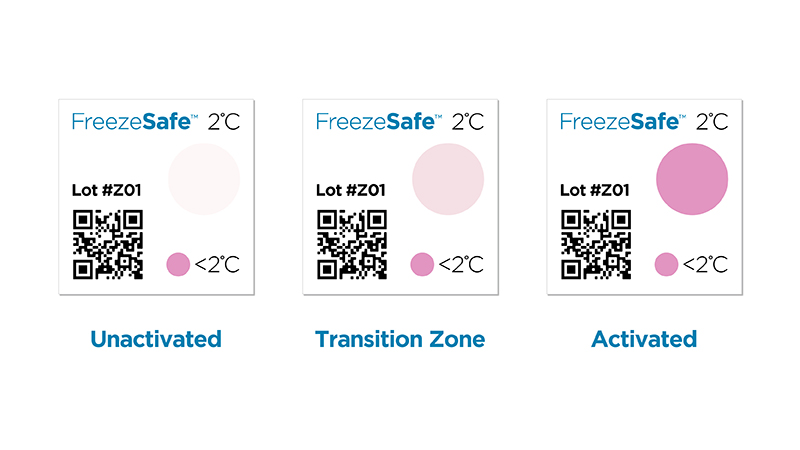
FreezeSafe: Low-Cost Simple Indicator for Life Science Application
Safeguarding Pharmaceutical Integrity: How FreezeSafe from SpotSee Ensures Vital Temperature Control In the world of pharmaceuticals, maintaining temperature control is paramount to preserving the efficacy and safety of various products. Certain pharmaceuticals, such...
Blood Monitoring Solutions
Cold chain logistics is the larger system and equipment used to safely transport goods that are temperature sensitive. Think of the cold supply chain as consistent and uninterrupted temperature control throughout the entire supply chain network.